It is known that premenopausal women have a
lower risk of heart attacks and cancer than do men of the same age.
The advantage is partially due to the monthly loss of iron in
the menses. This is confirmed by the fact that
postmenopausal women lose this advantage, while men who donate blood
as little as once a year have the same reduced risk as premenopausal
women.
Chelation has an affinity for iron
as shown on the chart two pages back. If you
look on the chart, you will see that
EDTA can
remove two different forms of iron, both the metabolically
active Fe2+ and the oxidative Fe3+ forms of iron, but that it
preferentially bonds to the oxidative Fe3+ form as evidences by Fe3+
being to the left of Fe2+ on the chart. This is
an important distinction, as you will see, since Fe3+ is quite a
problem in the body.
Methemoglobinemia is the clinical state in which circulating
hemoglobin is present with iron in the oxidized Fe3+ state instead
of the usual Fe2+ state. Fe3+ cannot carry
oxygen. Fe 3+ also moves the oxyhemoglobin
dissociation curve to the left, thus what little oxygen is present
on other hemoglobin molecules has a harder time being released into
the tissues. The lack of oxygen is obvious from
the chocolate brown appearance (shown below) of methemoglobineminic
blood.
In health, a small amount of oxidized Fe3+ iron is formed by
auto-oxidation of the circulating red blood cells.
Fe2+ is oxidized to Fe3+ at the rate of about 3% per day.
This iron is reduced back to Fe2+ by the enzyme NADH-cytochrome b5
reductase and reduced glutathione.
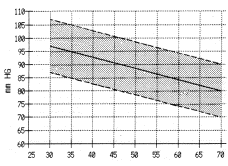
Perhaps the gradual decrease of
tissue oxygen seen as we age (see chart below) is due to the
downgrading of these antioxidant systems. While
enzyme NADH-cytochrome b5 reductase may be upregulated with niacin,
this enzyme accounts for perhaps only 5% of the reduction of Fe3+ to
Fe2+. Glutathione reduces Fe3+ to Fe2+ non-enzymatically,
but it is a difficult antioxidant to raise in the body.
Perhaps our best defense against Fe3+ for now is the use of
EDTA.
Since local anesthetics cause the
oxidation of Fe2+ to Fe3, and I.V. chelation is usually given with
procaine (to numb the burning sensation caused by Di-sodium EDTA),
we see another advantage of suppository chelation with Magnesium di-potassium
EDTA, over I.V. chelation, in that it will not cause the conversion
of Fe2+ to Fe3+ thus lowering oxygen carrying and delivery capacity
since no anesthetics are required.

Return to Medicardium Main Information Page
Medicardium Protocol & Suggested Use
Medicardium
EDTA
Research Studies
Medicardium Ingredients & Instructions